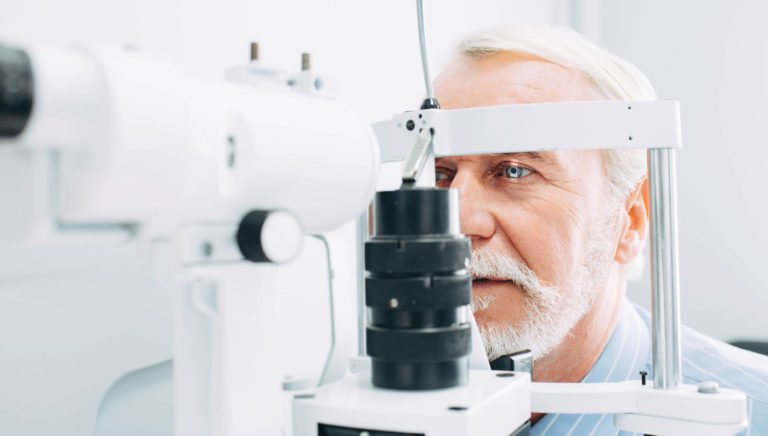
Enrolling Studies
Keystone Research is best known for ophthalmology clinical studies in Austin. We specialize in conducting clinical trials for Glaucoma and Ocular Hypertension and are also actively involved in Dry Eye clinical trials.

Participant Info
Participate in the ophthalmology clinical research in Austin and receive compensation for your time and travel. No insurance or proof of residency is required. You get advanced medication for your condition, health exams and medical reports.

Sponsor/CRO Info
Keystone Research has over 350 completed clinical trials in the past three decades. We collaborate with industry leaders to bring FDA-approved treatment, in pharmaceuticals, devices, and equipment.
Benefits of Volunteering in Clinical Studies in Austin, TX
Keystone Research is dedicated to advancing ophthalmology clinical trials in Austin by bridging the gap between the industry and sight-saving innovations. Here are the key benefits of participating in our paid clinical studies in Austin, TX.
Early Access to Advanced Ophthalmic Innovations
Our ophthalmology clinical studies allow access to groundbreaking innovations in medicine. By volunteering for these studies, participants become part of the journey toward FDA-approved, sight-saving advancements.
Expert Medical Guidance
Our paid clinical trials in Austin are led by experienced medical professionals and researchers, specialized in the ophthalmology field. We ensure that participants will receive high-quality medical care throughout the study.
Contribution to Medical Advancements
Our paid research studies in Austin contribute to the development of new drugs, therapies, and medical technologies. We help participants understand diseases better and refine treatment approaches.
Expertise in All Trial Phases
Why Choose Keystone Research For Ophthalmology Clinical Trials, Austin?
Keystone Research can be a trusted ally in advancing ophthalmology research and studies in Austin. Here’s why you should participate in clinical trials.
Advanced Facilities
Our research facilities are equipped with the latest technologies and resources necessary for successful Ophthalmology Clinical Trials in Austin.
Experienced Investigator Team
Safe & Sound research
Informed consent
Your participation in our clinical trial will always be voluntary, and you will never be enrolled without your explicit knowledge and consent. We educate about the potential risks and benefits of the study
Participate in Ophthalmology Clinical Studies in Austin, TX
Frequently Asked Questions
At Keystone Research, we follow a systematic approach to assess the safety and effectiveness of potential treatments. The ophthalmology studies typically involve the following activities:
- Pre-Screening
- Informed Consent
- Study Visits (Treatment Visits)
- End of Study Visit
- Follow-up Safety Visit
At Keystone Research, the appointment visit generally lasts at least 30 minutes and may extend to a few hours.
Participation is open to individuals who meet specific eligibility criteria outlined by the study protocol. Eligibility factors may include age, gender, health condition, and other relevant criteria depending on the nature of the research. Consult with our research team to determine your eligibility and suitability for a particular study.
At Keystone Research, we follow a research plan, referred to as a study protocol to address specific research inquiries and ensure participant safety. This protocol encompasses key details such as
- The study’s purpose.
- Inclusion and exclusion criteria specifying who can participate.
- The required number of participants.
- A schedule outlining tests, procedures, investigational product details, and dosages.
- The overall duration of the study.
- The information to be collected about the participants.
To get started, reach out to our experts or complete the form available on our website. Our experts will get back to you with a list of clinical trials open for participation.
